DHEA与抗衰老(专业版)
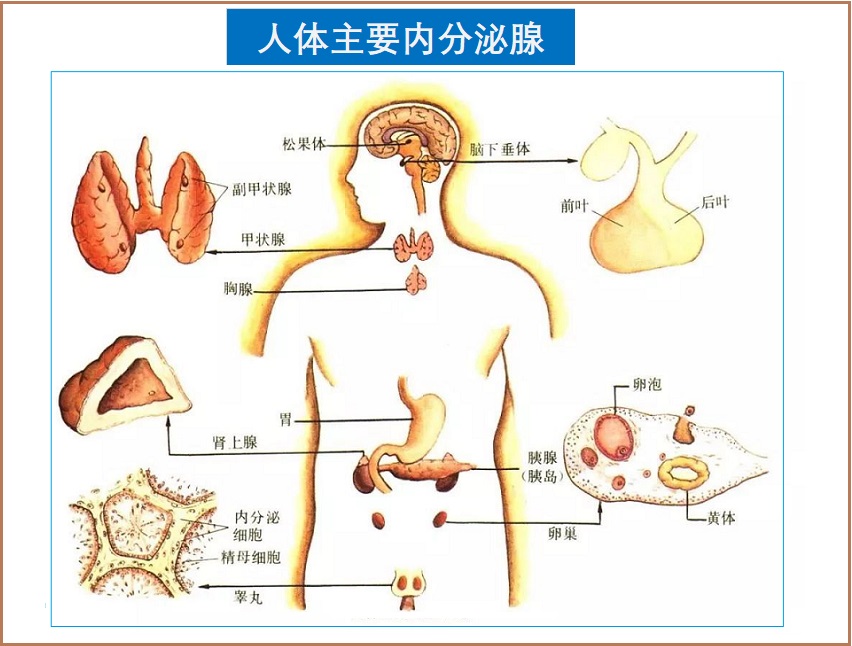
作为雄激素和雌激素的前体物,脱氢表雄酮(DHEA)在维持荷尔蒙平衡和青春活力方面发挥着重要作用。此外,DHEA可直接作用和调节涉及身体健康和疾病各个方面的途径。
其他名称:DHEA恢复疗法
英文名称:DHEA Restoration Therapy
衰老会破坏荷尔蒙平衡,与年轻人相比,几种关键荷尔蒙的水平显著降低,DHEA也不例外。到80岁时,DHEA水平与年轻成年期相比下降了80%-90%3。在了解了DHEA在支持多个身体系统的健康、年轻生理学方面所起的作用后,这一问题的严重性就变得很清楚了。研究表明,DHEA-S水平的降低与许多年龄相关疾病状态的病理生理学有关,包括认知能力下降、心血管疾病、骨质流失、癌症、抑郁症、性功能障碍和各种炎症性疾病3,1,2,4-8。
恢复年轻人的DHEA水平提供了一个独特的机会来减轻激素水平下降的后果。与雄激素(即睾酮替代疗法)或雌激素(即雌激素替代疗法)的直接给药不同,提高DHEA水平提供了这种激素前体的“储备池”,各种组织可以将其转化为雄激素和雌激素1,3,9-11。然而,DHEA给药不能取代测量和恢复其他激素的需要,因为它转化为雄激素和雌激素的速率因个体和性别而异3,9,13-15。因此,恢复DHEA水平应被视为综合激素恢复方案的一个组成部分,而不是男性睾酮替代和女性雌激素替代的替代方案。
由于DHEA-S在血液中比DHEA更丰富1,2,测量DHEA-S浓度的简单血液测试可以整合到任何男性和女性的健康衰老策略中。通过定期监测DHEA-S和其他激素的血液水平,为个体提供关于其激素环境状态的特定反馈1。这允许开发、实施和优化个性化方案,帮助中老年人过上充实、活跃、健康的生活3。
生物相同激素替代疗法(Bioidentical HRT)是一种给予与人体产生的激素结构相同的激素的方法。DHEA治疗也是生物相同的,是任何综合激素恢复方案的组成部分。另一方面,一些形式的常规激素替代疗法使用与人类产生的激素不同的激素,这些激素要么来源于动物,要么是合成的。有证据表明,与传统的激素替代疗法相比,生物相同的激素替代治疗可能更安全,并与更高的患者满意度相关16。
更多关于女性、男性荷尔蒙恢复/平衡内容,可参阅本网相关专文:
DHEA产生的主要位置是肾上腺的外层,称为肾上腺皮质;一些其他组织,如男性的睾丸和绝经前女性的卵巢,以及大脑等也会产生DHEA17,但程度要小得多。DHEA的产生在生命的第二到第三个十年达到峰值。此后,水平随着年龄的增长而稳步下降1,3。
除了作为激素前体的作用外,DHEA还调节炎症,这是许多慢性疾病的驱动因素。这种多功能激素还通过激活一种名为内皮一氧化氮合酶(eNOS)的酶,促进血管内皮细胞信号分子一氧化氮(NO)的产生。NO通过其刺激血管扩张的能力,是血液流动的关键调节因子。因此,在医学文献中,低DHEA水平与心血管疾病有关并不奇怪3,1。
口服后,DHEA主要转化为DHEA-S,DHEA-S在血液中循环的时间远长于DHEA。循环DHEA-S起着组织可以利用的储备作用。一旦被组织吸收,DHEA-S被转换回DHEA,然后DHEA可以局部转化为雄激素和雌激素或发挥直接作用3,1。
直到21世纪初,许多关于DHEA的研究都集中在它作为雄激素和雌激素的前体的作用上。然而,最近的研究揭示了DHEA直接介导的几种生物学作用。研究表明,血管内膜(内皮)、心脏、肾脏和肝脏细胞膜上的特异性受体与DHEA直接相互作用3,1。例如,DHEA的一个重要的雄激素和雌激素非依赖性作用是激活血管中一种名为内皮一氧化氮合酶(eNOS)的酶,该酶产生对健康血管功能重要的强效血管舒张剂一氧化氮(NO)3,1,18-20。
尽管肾上腺产生了大部分DHEA,但它也可以由大脑产生21,17。此外,中枢神经系统(CNS)中的DHEA水平是血液中的6-8倍1。这导致几位研究人员将DHEA归类为“神经甾体”21,22。DHEA已被证明可以调节大脑各个区域神经递质的释放和信号传导。因此,DHEA对某些涉及大脑的健康状况,如抑郁症和焦虑症,引起了人们的兴趣,这并不奇怪1,3,4。
随着人类年龄的增长,认知功能和记忆通常会受损。这和与年龄相关的大脑神经类固醇水平下降相对应。同样,一些神经退行性疾病,如阿尔茨海默病,也与神经甾体水平下降有关11,23。人们认为,与年龄相关的DHEA下降可能会损害神经元功能和完整性23。
几项研究揭示了DHEA与各种环境下认知功能之间的关系。一项对755名老年人进行了3年跟踪调查的研究发现,根据迷你精神状态检查(MMSE)(一种标准化的认知评估)测量,DHEA-S水平随着认知功能的下降而下降。此外,在基线MMSE中得分较高的受试者比得分较差的受试对象更有可能具有更高的DHEA-S水平,并且在基线时具有较低的DHEA-S-水平可以预测在研究期间认知功能的更大下降24。在另一项针对24名健康年轻男性的研究中,DHEA剂量为150mg,每天两次,持续7天,可改善情绪和记忆力。这项研究还发现,补充DHEA可以降低皮质醇的夜间水平,皮质醇是一种在压力下释放的激素25。一项单独的双盲、安慰剂对照研究招募了24名绝经后妇女,通过几项标准化测试,发现每天50mg的DHEA可以改善视觉空间表现。研究人员还发现,更高水平的DHEA及其代谢物与更好的视觉空间任务表现相关26。一项针对居住在日本辅助护理机构的27名65-90岁女性的研究发现,在6个月内每天补充25mg DHEA可提高接受积极治疗的受试者的认知得分,而接受安慰剂治疗的受测者的认知功能则有所恶化27。
DHEA调节某些人群认知功能的一种方法是保留几种神经保护因子的产生,如IGF-1(胰岛素样生长因子-1)、VEGF(血管内皮生长因子)和TGF-β(转化生长因子β)。一项实验室研究测量了从轻度至中度阿尔茨海默病患者身上提取的细胞产生的这些神经保护因子的水平,并将结果与从健康、年龄匹配的对照组中提取的样本进行了比较。科学家们发现,与健康细胞相比,阿尔茨海默病患者的细胞产生的这些神经保护生长因子的量显著减少。然而,当阿尔茨海默症患者的细胞与DHEA-s孵育时,生长因子的产生恢复到与健康对照细胞相似的水平。作者评论道:“这些数据表明,DHEA-S能够增加……神经保护性生长因子的产生……这为治疗痴呆症提供了一种新的方法”28。
此外,DHEA可能通过对抗糖皮质激素(如皮质醇)对神经元的有害影响而发挥神经保护作用。这是情绪障碍的一个重要考虑因素,因为糖皮质激素升高与社交焦虑和抑郁等精神疾病有关29。事实上,研究已经将抑郁症与成人、老年人和青少年人群的低血清DHEA浓度联系起来29,30,5。补充DHEA也与减少精神分裂症患者的焦虑和改善对抗精神病药物的反应有关31,32。在中年人中,DHEA(每天90mg,持续3周,然后每天450mg,持续三周)改善了恶劣心境障碍,这是一种慢性、轻度抑郁情绪33。在6个月的时间里,每天补充50mg的DHEA可以改善老年男性和女性的心理健康10。另一项研究发现,DHEA每天100–400mg,持续8周,可缓解HIV/AIDS患者的非严重、持续的抑郁症34。当垂体功能不理想时,DHEA水平的恢复也可能支持情绪。每天50mg的DHEA替代治疗使接受生长激素替代治疗的男性和女性垂体功能减退患者的心理健康状况得到长期改善35。
2. DHEA与骨骼健康
尽管人们通常认为骨质疏松症只影响女性,但它会影响男性的生活。在美国,数百万男性受到骨质疏松症或低骨量的影响,而且这个数字可能会随着人口年龄的增长而增长36-38。此外,有证据表明,与DHEA-S水平正常的绝经后妇女相比,骨密度低的绝经后女性的DHEA-S含量较低。事实上,捷克的一项研究发现,86%的DHEA-S水平处于分布较低的四分之一的女性骨密度较低,而健康绝经后女性的骨密度预计约为30%39。骨密度主要由两种细胞类型调节:造骨细胞和破骨细胞,前者构建骨骼,后者分解或吸收骨骼。DHEA促进成骨细胞活性并抑制破骨细胞介导的骨破坏。它似乎通过转化为刺激成骨细胞活性的雌激素,以及通过雄激素和雌激素非依赖性机制来实现这一点40,41。
骨组织对激素调节特别敏感。因此,与年龄相关的激素水平下降,包括DHEA,对男性和女性的骨骼健康都有相当大的影响42,43。DHEA和其他雄性激素在骨形成过程中起着关键作用;因此,DHEA水平下降可能会损害骨代谢并促进骨质疏松症40,3。DHEA已被证明可以有效治疗骨质疏松症,在其他方面健康的老年妇女中,持续一年每天50mg的剂量可以增加腰椎的骨矿物质密度。DHEA还显示出降低血清中1型胶原的C末端肽,这是骨转换的标志物44,45。在另一项研究中,65-75岁的女性每天服用50mg DHEA、650IU维生素D和700mg钙,治疗2年后,脊柱骨密度增加了3.6%43。
3. DHEA与心血管健康
与衰老相关的DHEA-S的下降可能会导致血管疾病和心脏事件的风险,尤其是在绝经后妇女中46。在男性中,DHEA-S水平降低似乎与糖尿病和冠心病的高风险相关47。观察性研究还表明,随着DHEA-S水平的下降,心血管疾病发病率上升48。
补充DHEA已被证明可以改善心血管健康。在健康的老年受试者中,DHEA的短期治疗似乎增加了NO的产生,降低了低密度脂蛋白(LDL)胆固醇,并提高了睾酮水平49。还发现DHEA可抑制血管最内层细胞(内皮)的炎症过程50。肥胖女性在3个月内服用100mg DHEA-S后,脂肪酸平衡发生了变化,血液中的饱和脂肪减少,从而表明其代谢状况更健康51,52。
此外,DHEA可能支持损伤后血管组织的健康重塑50。在接受心血管手术(即冠状动脉造影)的女性中,在6年的随访时间内,DHEA-S水平处于分布的后三分之一的女性比处于分布的前三分之二的女性更有可能死于任何原因。具体而言,尽管21%的DHEA-S水平处于分布的后三分之一的女性在随访期间死亡,但只有10%的DHEA-S-水平处于分布前三分之二的女性死亡。这一证据表明DHEA在心脏病中具有进一步的保护作用53。动物研究进一步表明,DHEA对血管重塑存在有利作用54。
4. DHEA与血糖调节
DHEA似乎可以增加胰岛素敏感性并对抗胰岛素抵抗。胰岛素抵抗是2型糖尿病的早期指标,与肥胖密切相关,两者都是心脏病的主要风险因素55,56。DHEA已被证明对糖尿病具有保护作用57。事实上,一项研究表明,口服糖耐量测试显示,服用50mg DHEA 持续1年可改善胰岛素反应;在研究开始时葡萄糖耐量受损的参与者中,2年后进一步改善58。另一项研究表明,在6个月内每天服用50mg DHEA可降低胰岛素抵抗59。这些研究的综合结果可能表明,可能需要长期给药才能看到胰岛素抵抗的显著变化。
另一组研究人员发现,在12周内每天补充50mg DHEA后,肾上腺功能可能受损的女性表现出胰岛素敏感性的改善60。另一项研究发现,77%患有冠状动脉疾病的2型糖尿病男性DHEA-S水平较低。当低DHEA-S与其他三种风险因素(睾酮缺乏、hs-CRP升高和血清N-末端B型钠尿肽原升高)相结合时,心血管死亡风险比健康对照受试者高出惊人的63倍61。其他证据表明,DHEA可以防止高浓度葡萄糖引起的血管损伤57。血糖升高会通过一种称为糖基化的过程引起氧化应激和功能失调蛋白质的形成,从而造成损害。一项针对20名2型糖尿病患者的研究表明,连续12周每天补充50mg DHEA可以改善高血糖水平引起的氧化失衡,并防止晚期糖基化终产物(AGE)的形成。这些发现表明,DHEA可能对2型糖尿病患者慢性并发症的发作和/或进展产生有益影响62。
5.DHEA与免疫功能
DHEA的另一个重要作用是在免疫系统中对抗皮质醇(“应激激素”)的作用。当DHEA增强免疫力时,皮质醇会抑制它63,64。这对老年人来说尤其重要,因为年龄的增长与DHEA:皮质醇比率的降低有关64。换言之,老年人比年轻人更容易受到皮质醇的免疫抑制,这可能会增加他们的感染风险63。
事实上,人们认为,与年龄相关的DHEA缺乏,导致DHEA和皮质醇之间的失衡,可能是老年人常见的免疫功能下降的部分原因63-65。一般来说,随着年龄的增长,免疫系统的功能会下降。这被称为免疫衰老。同样,由于多个腺体(包括肾上腺皮质)功能丧失而导致的激素分泌下降被称为内分泌衰老(Endocrinosenescence)64。在老年人中,每天50mg的DHEA可以增强免疫系统,并有可能预防一些常见的感染65。
DHEA和皮肤衰老
DHEA已被证明在皮肤中具有抗氧化和抗炎作用66,67,DHEA的减少与皮肤萎缩和皮肤衰老增加有关68,69。局部施用DHEA可以通过刺激胶原蛋白生物合成和改善真皮(直接位于皮肤表面下的组织层)的结构组织,在皮肤中发挥抗衰老作用68,70。通过对绝经后4个月以上的妇女局部施用1%的DHEA配方,研究参与者体验到皮脂分泌增加,这有助于皮肤的柔软性71。
DHEA与男、女性功能
大量研究调查了DHEA与性健康之间的关系,尤其是在女性中。随着女性年龄的增长,除了骨质疏松症和心脏病的风险增加外,性功能和性欲也趋于下降72。DHEA已被证明可以改善性功能的几乎所有方面,包括欲望、性唤起、活动、兴趣和性欲1。一项研究发现,对有中度至重度阴道萎缩症状的绝经后妇女阴道给予DHEA,可对性功能的几个重要方面产生有益影响,包括性唤起/感觉、润滑和性高潮73。另一项研究发现,在健康的绝经后妇女中,每天口服10mg DHEA治疗可显著改善性功能和性交频率74。
DHEA也被证明有益于男性的性健康1。在6个月内,每天服用50mg DHEA的阳痿男性性功能得到改善,但PSA、睾酮、泌乳素或前列腺大小没有增加75。
DHEA和减肥(关于7-Keto®DHEA)
7-Keto®DHEA是DHEA的代谢产物,不会转化为睾酮或雌激素,但具有促荷尔蒙特性76,77。7-Keto®DHEA似乎增加了基础代谢率和产热(即将储存的能量转化为体内热量)78-80。更高的基础代谢率和产热会导致能量储存(即身体脂肪)的减少。
在一项研究中,使用含有50mg 7-Keto®DHEA以及柠檬酸钙、绿茶提取物、维生素C、铬和维生素D3的组合配方制剂,超重成年人的静息代谢率增加了3.4%81。此外,DHEA及其代谢物抵消皮质醇的作用,皮质醇是一种分解代谢应激激素,与更大的脂肪积累有关82-87。在健康男性中,7-Keto®DHEA已被证明是安全的,剂量高达200mg/天,持续4周88。
DHEA在炎症和自身免疫中的作用
随着人类年龄的增长,免疫系统会减弱。免疫系统这种逐渐衰老的可能后果之一是某些癌症的发病率和感染易感性增加89。DHEA调节免疫系统的几个方面。在老年男性中,DHEA似乎改善了细胞因子的产生和T细胞、B细胞、NK细胞和单核细胞的功能90。炎症标志物白细胞介素-6(IL-6)和肿瘤坏死因子-α(TNF-α)在慢性炎症或炎症性疾病患者中均升高,似乎受到DHEA的积极影响91-94。在一项实验研究中,从哮喘患者身上提取免疫细胞,并用DHEA进行治疗。科学家们发现,DHEA抑制了这些细胞炎症介质的释放,并降低了它们的高反应性95。
此外,研究表明,在某些情况下,恢复最佳DHEA水平可能起到免疫调节剂的作用96。一项研究显示,患有自身免疫性甲状腺功能减退症的女性受试者的DHEA和某些代谢产物减少97。另一项研究发现,一组女性1型糖尿病患者的DHEA-S降低98。根据对几项研究的全面综述,在系统性红斑狼疮中,DHEA导致生活质量的临床显著改善99。在成年女性中,用DHEA(每天200mg)治疗狼疮可使发作患者的数量减少16%,每天50-200mg的DHEA在临床上是有益的100,101。
DHEA与预防身体虚弱
一些研究将老年人DHEA的下降视为衰老的主要标志。一项这样的研究发现,DHEA的下降与生活质量指标的下降是一致的,比如女性步态速度、精神状态和神经心理学评分等。从根本上讲,DHEA-S水平最高的老年女性在这些测试中表现最好。因此,测量DHEA-S血液水平可能是帮助确定个人衰老率的一种简单方法102。
跌倒和骨折是老龄化人口的一大担忧。DHEA可能会在这方面提供帮助。在一项为期6个月的试验中,低DHEA-S、低骨密度和虚弱的女性每天服用50mg的DHEA(以及维生素D和钙),每周进行2次温和的运动。试验结束时,下肢肌肉力量和功能得到改善103。
DHEA与长寿
许多科学研究发现,DHEA水平下降与多种原因导致的死亡可能性增加有关:
有趣的是,除了DHEA-S的总体水平外,一些证据表明,它随着年龄的增长而下降的速度可能独立影响寿命。在一项针对950名65岁或65岁以上个体的研究中,那些DHEA-S水平下降幅度更大的受试者在研究期间死亡的可能性比DHEA-S下降幅度更慢的受试对象高75%。尽管本研究中基线DHEA-S与死亡率无关,但这些发现仍然存在116。
DHEA与癌症风险
由于DHEA可能会增加性激素水平,人们对其在患有或患有激素相关癌症的人中的使用表示担忧。迄今为止,没有任何研究可令人信服地表明,由于补充DHEA或孕烯醇酮,导致人类激素依赖性癌症风险增加1,7。
实际上,真相更加微妙。哈佛泌尿科医生Abraham Morgentaler和其他人的重要研究表明,低睾酮水平可能会增加前列腺癌的风险,尽管这是一个有争议的概念。Morgentaler本人已经成为在老年男性中补充睾酮的有力支持者。他还是一项研究的首席研究员,该研究表明,在大鼠中补充DHEA可以提高总睾酮水平,而不会对前列腺组织产生任何有害变化117。
类似的理论风险适用于乳腺癌。但是,在天然雌二醇和孕酮(DHEA和/或孕烯醇酮的天然产物)组合的大型研究中,没有证明患乳腺癌的风险增加。此外,单独使用天然孕酮可以降低患癌症风险。这再次表明,使用DHEA和孕烯醇酮等前体提高性激素水平是安全的。最近的一项动物研究证明了DHEA对肥胖大鼠的直接抗癌作用118。
任何已知患有任何类型癌症的个人在使用任何新的补充剂或药物(包括DHEA)前,应咨询其医生。
参考文献:
1. Traish AM et al. Dehydroepiandrosterone (DHEA)--a precursor steroid or an active hormone in human physiology. J Sex Med. 2011 Nov;8(11):2960-82; quiz 2983.
2. Savineau JP et al. Role of DHEA in cardiovascular diseases. Biochemical pharmacology. Mar 15 2013;85(6):718-726.
3. Samaras N et al. A review of age related dehydroepiandrosterone (DHEA) decline and its association with well-known geriatric syndromes. Is treatment beneficial? Rejuvenation research. 2013 Aug;16(4):285-94.
4. Dong Y et al. Dehydroepiandrosterone sulphate: action and mechanism in the brain. Journal of neuroendocrinology. Jan 2012;24(1):215-224.
5. Załuska M et al. [Dehydroepiandrosteron (DHEA) in the mechanisms of stress and depression]. Psychiatr Pol. 2009 May-Jun;43(3):263-74.
6. Straub RH et al. Replacement therapy with DHEA plus corticosteroids in patients with chronic inflammatory diseases--substitutes of adrenal and sex hormones. Zeitschrift fur Rheumatologie. 2000;59 Suppl 2:Ii/108-118.
7. Krysiak R et al. Current views on the role of dehydroepiandrosterone in physiology, pathology and therapy. Pol Merkur Lekarski. 2008 Jan;24(139):66-71.
8. Lopez-Marure R et al. Effects of dehydroepiandrosterone on proliferation, migration, and death of breast cancer cells. European journal of pharmacology. Jun 25 2011;660(2-3):268-274.
9. Arlt W et al. Oral dehydroepiandrosterone for adrenal androgen replacement: pharmacokinetics and peripheral conversion to androgens and estrogens in young healthy females after dexamethasone suppression. The Journal of clinical endocrinology and metabolism. Jun 1998;83(6):1928-1934.
10. Morales AJ et al. Effects of replacement dose of dehydroepiandrosterone in men and women of advancing age. J Clin Endocrinol Metab. 1994 Jun;78(6):1360-7.
11. Aldred S et al. Decreased dehydroepiandrosterone (DHEA) and dehydroepiandrosterone sulfate (DHEAS) concentrations in plasma of Alzheimer's disease (AD) patients. Arch Gerontol Geriatr. 2010 Jul-Aug;51(1):e16-8.
12. Panjari M et al. DHEA therapy for women: effect on sexual function and wellbeing. Human reproduction update. May-Jun 2007;13(3):239-248.
13. Schulze J et al. SULT2A1 Gene Copy Number Variation is Associated with Urinary Excretion Rate of Steroid Sulfates. Frontiers in endocrinology. 2013 Jul 12:4:88.
14. Fitzpatrick JL et al. Metabolism of DHEA by cytochromes P450 in rat and human liver microsomal fractions. Archives of biochemistry and biophysics. May 15 2001;389(2):278-287.
15. Miller KK et al. Stereo- and regioselectivity account for the diversity of dehydroepiandrosterone (DHEA) metabolites produced by liver microsomal cytochromes P450. Drug metabolism and disposition: the biological fate of chemicals. Mar 2004;32(3):305-313.
16. Holtorf K. The bioidentical hormone debate: are bioidentical hormones (estradiol, estriol, and progesterone) safer or more efficacious than commonly used synthetic versions in hormone replacement therapy? Postgrad Med. 2009;121(1): 73-85.
17. Zwain IH et al. Dehydroepiandrosterone: biosynthesis and metabolism in the brain. Endocrinology. 1999 Feb;140(2):880-7.
18. Liu D et al. Dehydroepiandrosterone activates endothelial cell nitric-oxide synthase by a specific plasma membrane receptor coupled to Galpha(i2,3). The Journal of biological chemistry. Jun 14 2002;277(24):21379-21388.
19. Liu D et al. Dehydroepiandrosterone stimulates nitric oxide release in vascular endothelial cells: evidence for a cell surface receptor. Steroids. Apr 2004;69(4):279-289.
20. Simoncini T et al. Dehydroepiandrosterone modulates endothelial nitric oxide synthesis via direct genomic and nongenomic mechanisms. Endocrinology. Aug 2003;144(8):3449-3455.
21. Lazaridis I et al. Neurosteroid dehydroepiandrosterone interacts with nerve growth factor (NGF) receptors, preventing neuronal apoptosis. PLoS biology. Apr 2011;9(4):e1001051.
22. Baulieu EE et al. Dehydroepiandrosterone (DHEA) and dehydroepiandrosterone sulfate (DHEAS) as neuroactive neurosteroids. Proceedings of the National Academy of Sciences of the United States of America. Apr 14 1998;95(8):4089-4091.
23. Charalampopoulos I et al. Neurosteroids as modulators of neurogenesis and neuronal survival. Trends Endocrinol Metab. 2008 Oct; 19(8):300-7.
24. Valenti G et al. Dehydroepiandrosterone and cognitive function in the elderly: The InCHIANTI Study. J Endocrinol Invest. 2009 Oct; 32(9):766-72.
25. Alhaj HA et al. Effects of DHEA administration on episodic memory, cortisol and mood in healthy young men: a double-blind, placebo-controlled study. Psychopharmacology (Berl). 2006 Nov; 188(4):541-51.
26. Stangl B et al. Administration of dehydroepiandrosterone (DHEA) enhances visual-spatial performance in postmenopausal women. Behav Neurosci. 2011 Oct;125(5):742-52.
27. Yamada S et al. Effects of dehydroepiandrosterone supplementation on cognitive function and activities of daily living in older women with mild to moderate cognitive impairment. Geriatr Gerontol Int. 2010 Oct;10(4):280-7.
28. Luppi C et al. Growth factors decrease in subjects with mild to moderate Alzheimer's disease (AD): potential correction with dehydroepiandrosterone-sulphate (DHEAS). Arch Gerontol Geriatr. 2009;49 Suppl 1:173-84.
29. Herbert J. Neurosteroids, brain damage, and mental illness. Exp Gerontol. 1998 Nov-Dec;33(7-8):713-27.
30. Wong SY et al. Low DHEAS levels are associated with depressive symptoms in elderly Chinese men: results from a large study. Asian J Androl. 2011 Nov;13(6):898-902.
31. Ritsner MS. The clinical and therapeutic potentials of dehydroepiandrosterone and pregnenolone in schizophrenia. Neuroscience. 2011 Sep 15; 191:91-100.
32. Strous RD et al. Hormonal profile effects following dehydroepiandrosterone (DHEA) administration to schizophrenic patients. Clin Neuropharmacol. 2005 Nov-Dec;28(6):265-9.
33. Bloch M et al. Dehydroepiandrosterone treatment of midlife dysthymia. Biol Psychiatry. 1999 Jun 15;45(12):1533-41.
34. Rabkin JG et al. Placebo-controlled trial of dehydroepiandrosterone (DHEA) for treatment of nonmajor depression in patients with HIV/AIDS. Am J Psychiatry. 2006 Jan; 163(1):59-66.
35. Brooke AM et al. Dehydroepiandrosterone improves psychological well-being in male and female hypopituitary patients on maintenance growth hormone replacement. J Clin Endocrinol Metab. 2006 Oct; 91(10):3773-9.
36. Cawthon PM. Gender Differences in Osteoporosis and Fractures. Clin Orthop Relat Res. 2011 Jul;469(7):1900-5
37. Kawate H et al. [Secondary osteoporosis UPDATE. Treatment of male osteoporosis. Testosterone replacement therapy etc]. Clin Calcium. 2010 May;20(5):744-51.
38. Nuti R et al. Bone fragility in men: where are we? J Endocrinol Invest. 2010;33(7 Suppl):33-8.
39. Fingerova H et al. [Reduced serum dehydroepiandrosterone levels in postmenopausal osteoporosis]. Ceska gynekologie / Ceska lekarska spolecnost J. Ev. Purkyne. Apr 1998;63(2):110-113.
40. Adachi M et al. Role of androgens and DHEA in bone metabolism, Clin Calcium 2006 Jan; 16(1):61-6.
41. Wang YD et al. Dehydroepiandrosterone indirectly inhibits human osteoclastic resorption via activating osteoblastic viability by the MAPK pathway. Chin Med J (Engl). 2012 Apr; 125(7):1230-5.
42. Corina M et al. Relationship between bone mineral density, weight, and estrogen levels in pre- and postmenopausal women. Rev Med Chir Soc Med Nat Iasi. 2012 Oct-Dec;116(4):946-50.
43. Weiss EP et al. Dehydroepiandrosterone replacement therapy in older adults: 1- and 2-y effects on bone. Am J Clin Nutr. 2009 May; 89(5): 1459–1467.
44. Okuno S et al. Serum levels of C-terminal telopeptide of type I collagen: a useful new marker of cortical bone loss in hemodialysis patients. Osteoporos Int. 2005 May;16(5):501-9.
45. von Mühlen D et al. Effect of dehydroepiandrosterone supplementation on bone mineral density, bone markers, and body composition in older adults: the DAWN trial. Osteoporos Int. 2008 May;19(5):699-707.
46. Shufelt C et al. DHEA-S levels and cardiovascular disease mortality in postmenopausal women: results from the National Institutes of Health--National Heart, Lung, and Blood Institute (NHLBI)-sponsored Women's Ischemia Syndrome Evaluation (WISE). J Clin Endocrinol Metab. 2010 Nov;95(11):4985-92.
47. Ponholzer A et al. Vascular risk factors and their association to serum androgen levels in a population-based cohort of 75-year-old men over 5 years: results of the VITA study. World J Urol. 2009 Apr;28(2):209-14.
48. Mitchell LE et al. Evidence for an association between dehydroepiandrosterone sulfate and nonfatal, premature myocardial infarction in males. Circulation. 1994 Jan; 89(1):89-93.
49. Martina V et al. Short-term dehydroepiandrosterone treatment increases platelet cGMP production in elderly male subjects. Clin Endocrinol (Oxf). 2006 Mar; 64(3):260-4.
50. Li Y et al. Dehydroepiandrosterone inhibits CD40/CD40L expression on human umbilical vein endothelial cells induced by interferon gamma. Int Immunopharmacol. 2009 Feb; 9(2):168-72
51. Gómez-Santos C et al. Differential effect of oral dehydroepiandrosterone-sulphate on metabolic syndrome features in pre- and postmenopausal obese women. Clin Endocrinol (Oxf). 2012 Oct;77(4):548-54.
52. Gómez-Santos C et al. Dehydroepiandrosterone-sulphate replacement improves the human plasma fatty acid profile in plasma of obese women. Steroids. Dec 11 2011;76(13):1425-1432.
53. Shufelt C et al. DHEA-S levels and cardiovascular disease mortality in postmenopausal women: results from the National Institutes of Health--National Heart, Lung, and Blood Institute (NHLBI)-sponsored Women's Ischemia Syndrome Evaluation (WISE). J Clin Endocrinol Metab. 2010 Nov;95(11):4985-92.
54. Dumas de la Roque E et al. Dehydroepiandrosterone: A new treatment for vascular remodeling diseases including pulmonary arterial hypertension. Pharmacology & therapeutics. May 2010;126(2):186-199.
55. Basat O et al. Visceral adipose tissue and an indicator of insulin resistance in nonobese patients with new onset type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes. 2006;114(2):58-62.
56. Steinberger J et al. Obesity, Insulin Resistance, Diabetes, and Cardiovascular Risk in Children. Circulation. 2003;107:1448-53.
57. Heurta-Garcia E et al. Dehydroepiandrosterone inhibits the activation and dysfunction of endothelial cells induced by high glucose concentration. 2012 Feb;v.77(3),p.233-240.
58. Weiss EP et al. Dehydroepiandrosterone (DHEA) replacement decreases insulin resistance and lowers inflammatory cytokines in aging humans. Aging (Albany NY). 2011 May; 3(5): 533–542.
59. Talaei A et al. The effect of dehydroepiandrosterone on insulin resistance in patients with impaired glucose tolerance. Hormones (Athens). 2010 Oct-Dec;9(4):326-31.
60. Dhatariya K et al. Effect of dehydroepiandrosterone replacement on insulin sensitivity and lipids in hypoadrenal women. Diabetes. 2005 Mar;54(3):765-9.
61. Ponikowska B et al. Gonadal and adrenal androgen deficiencies as independent predictors of increased cardiovascular mortality in men with type II diabetes mellitus and stable coronary artery disease. Int J Cardiol. 2010 Sep 3;143(3):343-8.
62. Brignardello E et al. Dehydroepiandrosterone administration counteracts oxidative imbalance and advanced glycation end product formation in type 2 diabetic patients. Diabetes Care. 2007 Nov; 30(11):2922-7.
63. Butcher SK et al. Raised cortisol: DHEAS ratios in the elderly after injury: potential impact upon neutrophil function and immunity. Aging cell. Dec 2005;4(6):319-324.
64. Buford TW et al. Impact of DHEA(S) and cortisol on immune function in aging: a brief review. Appl Physiol Nutr Metab. 2008 Jun;33(3):429-33.
65. Roxas M et al. Colds and influenza: a review of diagnosis and conventional, botanical, and nutritional considerations. Altern Med Rev. 2007 Mar;12(1):25-48.
66. Puizina-Ivić N et al. Modern approach to topical treatment of aging skin. Coll Antropol. 2010 Sep;34(3):1145-53.
67. Chan C et al. Effect of dehydroepiandrosterone on atopic dermatitis-like skin lesions induced by 1-chloro-2,4-dinitrobenzene in mouse. Journal of dermatological science. Nov 01 2013.
68. El-Alfy M et al. Skin responses to topical dehydroepiandrosterone: implications in antiageing treatment? Br J Dermatol. 2010 Nov;163(5):968-76.
69. Labrie F. DHEA, important source of sex steroids in men and even more in women. Prog Brain Res. 2010;182:97-148.
70. Calvo E et al. Pangenomic changes induced by DHEA in the skin of postmenopausal women. J Steroid Biochem Mol Biol. 2008 Dec; 112(4-5):186-93.
71. Nouveau S et al. Effects of topical DHEA on aging skin: a pilot study. Maturitas. 2008 Feb 20;59(2):174-81.
72. Yasui T et al. Androgen in postmenopausal women. J Med Invest. 2012;59(1-2):12-27.
73. Labrie F et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause. 2009 Sep-Oct; 16(5):923-31
74. Genazzani AR et al. Effect of 1-year, low-dose DHEA therapy on climacteric symptoms and female sexuality. Climacteric. 2011 Dec;14(6):661-8.
75. Reiter WJ et al. Dehydroepiandrosterone in the treatment of erectile dysfunction: a prospective, double-blind, randomized, placebo-controlled study. Urology. 1999 Mar;53(3):590-4; discussion 594-5.
76. Worrel ME et al. Effects of 7-keto dehydroepiandrosterone on voluntary ethanol intake in male rats. Alcohol (Fayetteville, N.Y.). Jun 2011;45(4):349-354.
77. Amato RJ et al. A comparison of dehydroepiandrosterone and 7-keto dehydroepiandrosterone with other drugs that modulate ethanol intake in rats responding under a multiple schedule. Behavioural pharmacology. Jun 2012;23(3):250-261.
78. Bobyleva V et al. The effects of the ergosteroid 7-oxo-dehydroepiandrosterone on mitochondrial membrane potential: possible relationship to thermogenesis. Archives of biochemistry and biophysics. May 1 1997;341(1):122-128.
79. Ihler G et al. 7-oxo-DHEA and Raynaud's phenomenon. Medical hypotheses. Mar 2003;60(3):391-397.
80. Hampl R et al. Steroids and thermogenesis. Physiol Res. 2006; 55(2):123-31.
81. Zenk JL et al. HUM5007, a novel combination of thermogenic compounds, and 3-acetyl-7-oxo-dehydroepiandrosterone: each increases the resting metabolic rate of overweight adults. J Nutr Biochem. 2007 Sep; 18(9):629-34.
82. Moyer AE et al. Stress-induced cortisol response and fat distribution in women. Obesity research. May 1994;2(3):255-262.
83. Abraham SB et al. Cortisol, obesity, and the metabolic syndrome: a cross-sectional study of obese subjects and review of the literature. Obesity (Silver Spring, Md.). Jan 2013;21(1):E105-117.
84. Muller C et al. The native anti-glucocorticoid paradigm. J Steroid Biochem Mol Biol. 2006 Jul; 100(1-3):95-105.
85. Hennebert O et al. Dehydroepiandrosterone 7alpha-hydroxylation in human tissues: possible interference with type 1 11beta-hydroxysteroid dehydrogenase-mediated processes. J Steroid Biochem Mol Biol. 2007 May;104(3-5):326-33.
86. Marin P et al. Cortisol secretion in relation to body fat distribution in obese premenopausal women. Metabolism: clinical and experimental. Aug 1992;41(8):882-886.
87. Buoso E et al. Opposing effects of cortisol and dehydroepiandrosterone on the expression of the receptor for Activated C Kinase 1: implications in immunosenescence. Experimental gerontology. Nov 2011;46(11):877-883.
88. Davidson M et al. Safety and pharmacokinetic study with escalating doses of 3-acetyl-7-oxo-dehydroepiandrosterone in healthy male volunteers. Clin Invest Med. 2000 Oct;23(5):300-10.
89. Ramos-Casals M et al. Autoimmunity and geriatrics: clinical significance of autoimmune manifestations in the elderly. Lupus. 2003;12(5):341-55.
90. Khorram O et al. Activation of immune function by dehydroepiandrosterone (DHEA) in age-advanced men. J Gerontol A Biol Sci Med Sci . 1997 Jan;52(1):M1-M7.
91. James K et al. IL-6, DHEA and the ageing process. Mech Ageing Dev . 1997 Feb;93(1-3):15-24.
92. Straub RH et al. Serum dehydroepiandrosterone (DHEA) and DHEA sulfate are negatively correlated with serum interleukin-6 (IL-6), and DHEA inhibits IL-6 secretion from mononuclear cells in man in vitro: possible link between endocrinosenescence and immunosenescence. J Clin Endocrinol Metab . 1998 Jun;83(6):2012-7.
93. Straub RH et al. Dehydroepiandrosterone in relation to other adrenal hormones during an acute inflammatory stressful disease state compared with chronic inflammatory disease: role of interleukin-6 and tumour necrosis factor. Eur J Endocrinol . 2002 Mar;146(3):365-74.
94. Leowattana W. DHEA(S): the fountain of youth. J Med Assoc Thai. 2001 Oct;84(Suppl 2):S605-S612.
95. Choi IS et al. Effects of dehydroepiandrosterone on Th2 cytokine production in peripheral blood mononuclear cells from asthmatics. Korean J Intern Med. 2008 Dec; 23(4):176-81.
96. Hazeldine J et al. Dehydroepiandrosterone as a regulator of immune cell function. J Steroid Biochem Mol Biol. 2010 May 31;120(2-3):127-36.
97. Drbalová K et al. Immunoprotective steroids and SHBG in non-treated hypothyroidism and their relationship to autoimmune thyroid disorders. Physiol Res. 2008;57 Suppl 1:S119-25.
98. Simunkova K et al. Adrenocortical function in young adults with diabetes mellitus type 1. J Steroid Biochem Mol Biol. 2010 Oct;122(1-3):35-41.
99. Crosbie D et al. Dehydroepiandrosterone for systemic lupus erythematosus. Cochrane Database Syst Rev. 2007 Oct 17;(4):CD005114.
100. van Vollenhoven RF et al. Treatment of systemic lupus erythematosus with dehydroepiandrosterone: 50 patients treated up to 12 months. J Rheumatol . 1998 Feb;25(2):285-9.
101. Chang DM et al. Dehydroepiandrosterone treatment of women with mild-to-moderate systemic lupus erythematosus: a multicenter randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2002 Nov;46(11):2924-7.
102. Sanders JL et al. Concurrent change in dehydroepiandrosterone sulfate and functional performance in the oldest old: results from the Cardiovascular Health Study All Stars study. The journals of gerontology. Series A, Biological sciences and medical sciences. Sep 2010;65(9):976-981.
103. Kenny AM et al. Dehydroepiandrosterone combined with exercise improves muscle strength and physical function in frail older women. J Am Geriatr Soc. 2010 Sep;58(9):1707-14.
104. Barrett-Connor E et al. A prospective study of dehydroepiandrosterone sulfate, mortality, and cardiovascular disease. The New England journal of medicine. Dec 11 1986;315(24):1519-1524.
105. Trivedi DP et al. Dehydroepiandrosterone sulfate and mortality in elderly men and women. The Journal of clinical endocrinology and metabolism. Sep 2001;86(9):4171-4177.
106. Hsu HJ et al. Low plasma DHEA-S increases mortality risk among male hemodialysis patients. Experimental gerontology. Dec 2012;47(12):950-957.
107. Kakiya R et al. Decreased serum adrenal androgen dehydroepiandrosterone sulfate and mortality in hemodialysis patients. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association. Oct 2012;27(10):3915-3922.
108. Ohlsson C et al. Low serum levels of dehydroepiandrosterone sulfate predict all-cause and cardiovascular mortality in elderly Swedish men. The Journal of clinical endocrinology and metabolism. Sep 2010;95(9):4406-4414.
109. Phillips AC et al. Cortisol, DHEA sulphate, their ratio, and all-cause and cause-specific mortality in the Vietnam Experience Study. European journal of endocrinology. Aug 2010;163(2):285-292.
110. Enomoto M et al. Serum dehydroepiandrosterone sulfate levels predict longevity in men: 27-year follow-up study in a community-based cohort (Tanushimaru study). Journal of the American Geriatrics Society. Jun 2008;56(6):994-998.
111. Glei DA et al. Dehydroepiandrosterone sulfate (DHEAS) and risk for mortality among older Taiwanese. Annals of epidemiology. Jul 2006;16(7):510-515.
112. Mazat L et al. Prospective measurements of dehydroepiandrosterone sulfate in a cohort of elderly subjects: relationship to gender, subjective health, smoking habits, and 10-year mortality. Proceedings of the National Academy of Sciences of the United States of America. Jul 3 2001;98(14):8145-8150.
113. Jansson JH et al. von Willebrand factor, tissue plasminogen activator, and dehydroepiandrosterone sulphate predict cardiovascular death in a 10 year follow up of survivors of acute myocardial infarction. Heart (British Cardiac Society). Oct 1998;80(4):334-337.
114. Berr C et al. Relationships of dehydroepiandrosterone sulfate in the elderly with functional, psychological, and mental status, and short-term mortality: a French community-based study. Proceedings of the National Academy of Sciences of the United States of America. Nov 12 1996;93(23):13410-13415.
115. Ponikowska B et al. Gonadal and adrenal androgen deficiencies as independent predictors of increased cardiovascular mortality in men with type II diabetes mellitus and stable coronary artery disease. Int J Cardiol. 2010 Sep 3;143(3):343-8.
116. Cappola AR et al. Trajectories of dehydroepiandrosterone sulfate predict mortality in older adults: the cardiovascular health study. The journals of gerontology. Series A, Biological sciences and medical sciences. Dec 2009;64(12):1268-1274.
117. Rhoden EL et al. Effects of chronic administration of dehydroepiandrosterone on serum testosterone levels and prostatic tissue in rats. The Journal of urology. Nov 2003;170(5):2101-2103.
118. Hakkak R et al. Dehydroepiandrosterone intake protects against 7,12-dimethylbenz(a)anthracene-induced mammary tumor development in the obese Zucker rat model. Oncology reports. Aug 2010;24(2):357-362.
英文名称:DHEA Restoration Therapy
概述
作为雄激素和雌激素的前体物,脱氢表雄酮(DHEA)在维持荷尔蒙平衡和青春活力方面发挥着重要作用。此外,DHEA可直接作用和调节涉及身体健康和疾病各个方面的途径。人体大约在25岁开始,体内DHEA水平逐渐下降,补充DHEA可改善衰老性慢性疾病,包括认知/痴呆、心脑血管疾病、骨松症和性功能障碍等。在美国,DHEA既作为膳食补充剂也以药物形式出售。简介
脱氢表雄酮(DHEA)是人体内最多的循环天然类固醇激素,至今已有超过4000篇文献评估了其对身体许多不同细胞和组织的作用。这种多功能激素及其代谢产物硫酸脱氢表雄酮(DHEA-S)为男性和女性提供了关键的激素益处1,2。作为雄性激素和雌性激素的合成前体,DHEA在维持激素平衡和年轻活力方面发挥着重要作用。它还通过独立于其作为雄激素和雌激素前体的作用的直接作用,调节全身参与健康和疾病各个方面的多种途径3,1,2。衰老会破坏荷尔蒙平衡,与年轻人相比,几种关键荷尔蒙的水平显著降低,DHEA也不例外。到80岁时,DHEA水平与年轻成年期相比下降了80%-90%3。在了解了DHEA在支持多个身体系统的健康、年轻生理学方面所起的作用后,这一问题的严重性就变得很清楚了。研究表明,DHEA-S水平的降低与许多年龄相关疾病状态的病理生理学有关,包括认知能力下降、心血管疾病、骨质流失、癌症、抑郁症、性功能障碍和各种炎症性疾病3,1,2,4-8。
恢复年轻人的DHEA水平提供了一个独特的机会来减轻激素水平下降的后果。与雄激素(即睾酮替代疗法)或雌激素(即雌激素替代疗法)的直接给药不同,提高DHEA水平提供了这种激素前体的“储备池”,各种组织可以将其转化为雄激素和雌激素1,3,9-11。然而,DHEA给药不能取代测量和恢复其他激素的需要,因为它转化为雄激素和雌激素的速率因个体和性别而异3,9,13-15。因此,恢复DHEA水平应被视为综合激素恢复方案的一个组成部分,而不是男性睾酮替代和女性雌激素替代的替代方案。
由于DHEA-S在血液中比DHEA更丰富1,2,测量DHEA-S浓度的简单血液测试可以整合到任何男性和女性的健康衰老策略中。通过定期监测DHEA-S和其他激素的血液水平,为个体提供关于其激素环境状态的特定反馈1。这允许开发、实施和优化个性化方案,帮助中老年人过上充实、活跃、健康的生活3。
生物相同激素替代疗法(Bioidentical HRT)是一种给予与人体产生的激素结构相同的激素的方法。DHEA治疗也是生物相同的,是任何综合激素恢复方案的组成部分。另一方面,一些形式的常规激素替代疗法使用与人类产生的激素不同的激素,这些激素要么来源于动物,要么是合成的。有证据表明,与传统的激素替代疗法相比,生物相同的激素替代治疗可能更安全,并与更高的患者满意度相关16。
更多关于女性、男性荷尔蒙恢复/平衡内容,可参阅本网相关专文:
DHEA背景与生物学
人体通过两种酶反应从胆固醇中获得DHEA。首先,胆固醇被转化为孕烯醇酮,孕烯醇酮有时被称为“主激素”,因为它是激素级联的前体,最终产生主要的性激素—睾酮和雌激素。接下来,孕烯醇酮被转化为DHEA1-3。DHEA产生的主要位置是肾上腺的外层,称为肾上腺皮质;一些其他组织,如男性的睾丸和绝经前女性的卵巢,以及大脑等也会产生DHEA17,但程度要小得多。DHEA的产生在生命的第二到第三个十年达到峰值。此后,水平随着年龄的增长而稳步下降1,3。
除了作为激素前体的作用外,DHEA还调节炎症,这是许多慢性疾病的驱动因素。这种多功能激素还通过激活一种名为内皮一氧化氮合酶(eNOS)的酶,促进血管内皮细胞信号分子一氧化氮(NO)的产生。NO通过其刺激血管扩张的能力,是血液流动的关键调节因子。因此,在医学文献中,低DHEA水平与心血管疾病有关并不奇怪3,1。
口服后,DHEA主要转化为DHEA-S,DHEA-S在血液中循环的时间远长于DHEA。循环DHEA-S起着组织可以利用的储备作用。一旦被组织吸收,DHEA-S被转换回DHEA,然后DHEA可以局部转化为雄激素和雌激素或发挥直接作用3,1。
直到21世纪初,许多关于DHEA的研究都集中在它作为雄激素和雌激素的前体的作用上。然而,最近的研究揭示了DHEA直接介导的几种生物学作用。研究表明,血管内膜(内皮)、心脏、肾脏和肝脏细胞膜上的特异性受体与DHEA直接相互作用3,1。例如,DHEA的一个重要的雄激素和雌激素非依赖性作用是激活血管中一种名为内皮一氧化氮合酶(eNOS)的酶,该酶产生对健康血管功能重要的强效血管舒张剂一氧化氮(NO)3,1,18-20。
DHEA的作用
1.DHEA在情绪和大脑健康中的作用尽管肾上腺产生了大部分DHEA,但它也可以由大脑产生21,17。此外,中枢神经系统(CNS)中的DHEA水平是血液中的6-8倍1。这导致几位研究人员将DHEA归类为“神经甾体”21,22。DHEA已被证明可以调节大脑各个区域神经递质的释放和信号传导。因此,DHEA对某些涉及大脑的健康状况,如抑郁症和焦虑症,引起了人们的兴趣,这并不奇怪1,3,4。
随着人类年龄的增长,认知功能和记忆通常会受损。这和与年龄相关的大脑神经类固醇水平下降相对应。同样,一些神经退行性疾病,如阿尔茨海默病,也与神经甾体水平下降有关11,23。人们认为,与年龄相关的DHEA下降可能会损害神经元功能和完整性23。
几项研究揭示了DHEA与各种环境下认知功能之间的关系。一项对755名老年人进行了3年跟踪调查的研究发现,根据迷你精神状态检查(MMSE)(一种标准化的认知评估)测量,DHEA-S水平随着认知功能的下降而下降。此外,在基线MMSE中得分较高的受试者比得分较差的受试对象更有可能具有更高的DHEA-S水平,并且在基线时具有较低的DHEA-S-水平可以预测在研究期间认知功能的更大下降24。在另一项针对24名健康年轻男性的研究中,DHEA剂量为150mg,每天两次,持续7天,可改善情绪和记忆力。这项研究还发现,补充DHEA可以降低皮质醇的夜间水平,皮质醇是一种在压力下释放的激素25。一项单独的双盲、安慰剂对照研究招募了24名绝经后妇女,通过几项标准化测试,发现每天50mg的DHEA可以改善视觉空间表现。研究人员还发现,更高水平的DHEA及其代谢物与更好的视觉空间任务表现相关26。一项针对居住在日本辅助护理机构的27名65-90岁女性的研究发现,在6个月内每天补充25mg DHEA可提高接受积极治疗的受试者的认知得分,而接受安慰剂治疗的受测者的认知功能则有所恶化27。
DHEA调节某些人群认知功能的一种方法是保留几种神经保护因子的产生,如IGF-1(胰岛素样生长因子-1)、VEGF(血管内皮生长因子)和TGF-β(转化生长因子β)。一项实验室研究测量了从轻度至中度阿尔茨海默病患者身上提取的细胞产生的这些神经保护因子的水平,并将结果与从健康、年龄匹配的对照组中提取的样本进行了比较。科学家们发现,与健康细胞相比,阿尔茨海默病患者的细胞产生的这些神经保护生长因子的量显著减少。然而,当阿尔茨海默症患者的细胞与DHEA-s孵育时,生长因子的产生恢复到与健康对照细胞相似的水平。作者评论道:“这些数据表明,DHEA-S能够增加……神经保护性生长因子的产生……这为治疗痴呆症提供了一种新的方法”28。
此外,DHEA可能通过对抗糖皮质激素(如皮质醇)对神经元的有害影响而发挥神经保护作用。这是情绪障碍的一个重要考虑因素,因为糖皮质激素升高与社交焦虑和抑郁等精神疾病有关29。事实上,研究已经将抑郁症与成人、老年人和青少年人群的低血清DHEA浓度联系起来29,30,5。补充DHEA也与减少精神分裂症患者的焦虑和改善对抗精神病药物的反应有关31,32。在中年人中,DHEA(每天90mg,持续3周,然后每天450mg,持续三周)改善了恶劣心境障碍,这是一种慢性、轻度抑郁情绪33。在6个月的时间里,每天补充50mg的DHEA可以改善老年男性和女性的心理健康10。另一项研究发现,DHEA每天100–400mg,持续8周,可缓解HIV/AIDS患者的非严重、持续的抑郁症34。当垂体功能不理想时,DHEA水平的恢复也可能支持情绪。每天50mg的DHEA替代治疗使接受生长激素替代治疗的男性和女性垂体功能减退患者的心理健康状况得到长期改善35。
2. DHEA与骨骼健康
尽管人们通常认为骨质疏松症只影响女性,但它会影响男性的生活。在美国,数百万男性受到骨质疏松症或低骨量的影响,而且这个数字可能会随着人口年龄的增长而增长36-38。此外,有证据表明,与DHEA-S水平正常的绝经后妇女相比,骨密度低的绝经后女性的DHEA-S含量较低。事实上,捷克的一项研究发现,86%的DHEA-S水平处于分布较低的四分之一的女性骨密度较低,而健康绝经后女性的骨密度预计约为30%39。骨密度主要由两种细胞类型调节:造骨细胞和破骨细胞,前者构建骨骼,后者分解或吸收骨骼。DHEA促进成骨细胞活性并抑制破骨细胞介导的骨破坏。它似乎通过转化为刺激成骨细胞活性的雌激素,以及通过雄激素和雌激素非依赖性机制来实现这一点40,41。
骨组织对激素调节特别敏感。因此,与年龄相关的激素水平下降,包括DHEA,对男性和女性的骨骼健康都有相当大的影响42,43。DHEA和其他雄性激素在骨形成过程中起着关键作用;因此,DHEA水平下降可能会损害骨代谢并促进骨质疏松症40,3。DHEA已被证明可以有效治疗骨质疏松症,在其他方面健康的老年妇女中,持续一年每天50mg的剂量可以增加腰椎的骨矿物质密度。DHEA还显示出降低血清中1型胶原的C末端肽,这是骨转换的标志物44,45。在另一项研究中,65-75岁的女性每天服用50mg DHEA、650IU维生素D和700mg钙,治疗2年后,脊柱骨密度增加了3.6%43。
3. DHEA与心血管健康
与衰老相关的DHEA-S的下降可能会导致血管疾病和心脏事件的风险,尤其是在绝经后妇女中46。在男性中,DHEA-S水平降低似乎与糖尿病和冠心病的高风险相关47。观察性研究还表明,随着DHEA-S水平的下降,心血管疾病发病率上升48。
补充DHEA已被证明可以改善心血管健康。在健康的老年受试者中,DHEA的短期治疗似乎增加了NO的产生,降低了低密度脂蛋白(LDL)胆固醇,并提高了睾酮水平49。还发现DHEA可抑制血管最内层细胞(内皮)的炎症过程50。肥胖女性在3个月内服用100mg DHEA-S后,脂肪酸平衡发生了变化,血液中的饱和脂肪减少,从而表明其代谢状况更健康51,52。
此外,DHEA可能支持损伤后血管组织的健康重塑50。在接受心血管手术(即冠状动脉造影)的女性中,在6年的随访时间内,DHEA-S水平处于分布的后三分之一的女性比处于分布的前三分之二的女性更有可能死于任何原因。具体而言,尽管21%的DHEA-S水平处于分布的后三分之一的女性在随访期间死亡,但只有10%的DHEA-S-水平处于分布前三分之二的女性死亡。这一证据表明DHEA在心脏病中具有进一步的保护作用53。动物研究进一步表明,DHEA对血管重塑存在有利作用54。
4. DHEA与血糖调节
DHEA似乎可以增加胰岛素敏感性并对抗胰岛素抵抗。胰岛素抵抗是2型糖尿病的早期指标,与肥胖密切相关,两者都是心脏病的主要风险因素55,56。DHEA已被证明对糖尿病具有保护作用57。事实上,一项研究表明,口服糖耐量测试显示,服用50mg DHEA 持续1年可改善胰岛素反应;在研究开始时葡萄糖耐量受损的参与者中,2年后进一步改善58。另一项研究表明,在6个月内每天服用50mg DHEA可降低胰岛素抵抗59。这些研究的综合结果可能表明,可能需要长期给药才能看到胰岛素抵抗的显著变化。
另一组研究人员发现,在12周内每天补充50mg DHEA后,肾上腺功能可能受损的女性表现出胰岛素敏感性的改善60。另一项研究发现,77%患有冠状动脉疾病的2型糖尿病男性DHEA-S水平较低。当低DHEA-S与其他三种风险因素(睾酮缺乏、hs-CRP升高和血清N-末端B型钠尿肽原升高)相结合时,心血管死亡风险比健康对照受试者高出惊人的63倍61。其他证据表明,DHEA可以防止高浓度葡萄糖引起的血管损伤57。血糖升高会通过一种称为糖基化的过程引起氧化应激和功能失调蛋白质的形成,从而造成损害。一项针对20名2型糖尿病患者的研究表明,连续12周每天补充50mg DHEA可以改善高血糖水平引起的氧化失衡,并防止晚期糖基化终产物(AGE)的形成。这些发现表明,DHEA可能对2型糖尿病患者慢性并发症的发作和/或进展产生有益影响62。
5.DHEA与免疫功能
DHEA的另一个重要作用是在免疫系统中对抗皮质醇(“应激激素”)的作用。当DHEA增强免疫力时,皮质醇会抑制它63,64。这对老年人来说尤其重要,因为年龄的增长与DHEA:皮质醇比率的降低有关64。换言之,老年人比年轻人更容易受到皮质醇的免疫抑制,这可能会增加他们的感染风险63。
事实上,人们认为,与年龄相关的DHEA缺乏,导致DHEA和皮质醇之间的失衡,可能是老年人常见的免疫功能下降的部分原因63-65。一般来说,随着年龄的增长,免疫系统的功能会下降。这被称为免疫衰老。同样,由于多个腺体(包括肾上腺皮质)功能丧失而导致的激素分泌下降被称为内分泌衰老(Endocrinosenescence)64。在老年人中,每天50mg的DHEA可以增强免疫系统,并有可能预防一些常见的感染65。
DHEA和皮肤衰老
DHEA已被证明在皮肤中具有抗氧化和抗炎作用66,67,DHEA的减少与皮肤萎缩和皮肤衰老增加有关68,69。局部施用DHEA可以通过刺激胶原蛋白生物合成和改善真皮(直接位于皮肤表面下的组织层)的结构组织,在皮肤中发挥抗衰老作用68,70。通过对绝经后4个月以上的妇女局部施用1%的DHEA配方,研究参与者体验到皮脂分泌增加,这有助于皮肤的柔软性71。
DHEA与男、女性功能
大量研究调查了DHEA与性健康之间的关系,尤其是在女性中。随着女性年龄的增长,除了骨质疏松症和心脏病的风险增加外,性功能和性欲也趋于下降72。DHEA已被证明可以改善性功能的几乎所有方面,包括欲望、性唤起、活动、兴趣和性欲1。一项研究发现,对有中度至重度阴道萎缩症状的绝经后妇女阴道给予DHEA,可对性功能的几个重要方面产生有益影响,包括性唤起/感觉、润滑和性高潮73。另一项研究发现,在健康的绝经后妇女中,每天口服10mg DHEA治疗可显著改善性功能和性交频率74。
DHEA也被证明有益于男性的性健康1。在6个月内,每天服用50mg DHEA的阳痿男性性功能得到改善,但PSA、睾酮、泌乳素或前列腺大小没有增加75。
DHEA和减肥(关于7-Keto®DHEA)
7-Keto®DHEA是DHEA的代谢产物,不会转化为睾酮或雌激素,但具有促荷尔蒙特性76,77。7-Keto®DHEA似乎增加了基础代谢率和产热(即将储存的能量转化为体内热量)78-80。更高的基础代谢率和产热会导致能量储存(即身体脂肪)的减少。
在一项研究中,使用含有50mg 7-Keto®DHEA以及柠檬酸钙、绿茶提取物、维生素C、铬和维生素D3的组合配方制剂,超重成年人的静息代谢率增加了3.4%81。此外,DHEA及其代谢物抵消皮质醇的作用,皮质醇是一种分解代谢应激激素,与更大的脂肪积累有关82-87。在健康男性中,7-Keto®DHEA已被证明是安全的,剂量高达200mg/天,持续4周88。
DHEA在炎症和自身免疫中的作用
随着人类年龄的增长,免疫系统会减弱。免疫系统这种逐渐衰老的可能后果之一是某些癌症的发病率和感染易感性增加89。DHEA调节免疫系统的几个方面。在老年男性中,DHEA似乎改善了细胞因子的产生和T细胞、B细胞、NK细胞和单核细胞的功能90。炎症标志物白细胞介素-6(IL-6)和肿瘤坏死因子-α(TNF-α)在慢性炎症或炎症性疾病患者中均升高,似乎受到DHEA的积极影响91-94。在一项实验研究中,从哮喘患者身上提取免疫细胞,并用DHEA进行治疗。科学家们发现,DHEA抑制了这些细胞炎症介质的释放,并降低了它们的高反应性95。
此外,研究表明,在某些情况下,恢复最佳DHEA水平可能起到免疫调节剂的作用96。一项研究显示,患有自身免疫性甲状腺功能减退症的女性受试者的DHEA和某些代谢产物减少97。另一项研究发现,一组女性1型糖尿病患者的DHEA-S降低98。根据对几项研究的全面综述,在系统性红斑狼疮中,DHEA导致生活质量的临床显著改善99。在成年女性中,用DHEA(每天200mg)治疗狼疮可使发作患者的数量减少16%,每天50-200mg的DHEA在临床上是有益的100,101。
DHEA与预防身体虚弱
一些研究将老年人DHEA的下降视为衰老的主要标志。一项这样的研究发现,DHEA的下降与生活质量指标的下降是一致的,比如女性步态速度、精神状态和神经心理学评分等。从根本上讲,DHEA-S水平最高的老年女性在这些测试中表现最好。因此,测量DHEA-S血液水平可能是帮助确定个人衰老率的一种简单方法102。
跌倒和骨折是老龄化人口的一大担忧。DHEA可能会在这方面提供帮助。在一项为期6个月的试验中,低DHEA-S、低骨密度和虚弱的女性每天服用50mg的DHEA(以及维生素D和钙),每周进行2次温和的运动。试验结束时,下肢肌肉力量和功能得到改善103。
DHEA与长寿
许多科学研究发现,DHEA水平下降与多种原因导致的死亡可能性增加有关:
- 在一项针对270名疑似心肌血流量减少的女性的研究中,与DHEA水平降至分布的上三分之二的女性相比,DHEA-S水平低于分布的下三分之一的受试者在9年的随访期内死于任何原因的可能性高11%46。
- 在另一项研究中,242名年龄在50岁至79岁之间的男性被随访了12年,与较高水平相比,DHEA-S水平低于140µg/dL与心血管疾病死亡风险相关3.3倍。此外,即使在对一些混杂因素进行了调整后,DHEA-S中100µg/dL的增加也可使任何原因的死亡风险降低36%104。
- 剑桥大学的研究人员对963名男性进行了长达9年的跟踪调查,发现随着循环DHEA-S水平增加到分布的四分之一以上,任何原因导致的死亡风险都降低了约30%105。
- 在一项针对因慢性肾脏疾病接受透析的男性的研究中,与调整潜在混杂因素后的较高水平相比,低血浆DHEA-S水平与任何原因导致的死亡风险增加约2.9倍有关106。
- 对313名接受透析的男性进行的一项类似研究,证实了DHEA-S水平较低可预测该人群死亡率增加的发现107。
- 在一项对2644名瑞典男性进行平均4.5年随访的研究中,即使在研究调整了研究结果以考虑可能影响结果的变量后,DHEA-S水平低于分布的四分之一的男性在随访期间死亡的可能性比DHEA-S高的男性高54%108。
- 对4255名越南时代的美国退伍军人的分析显示,在15年的随访期内,较高的DHEA-S水平与49%的死亡可能性降低有关109。
- 一项对940名受试者进行了27年随访的长期研究发现,DHEA-S水平高于200µg/dL的男性在研究期间死亡的可能性明显低于水平较低的男性110。
- 一项对963名台湾老年人进行的为期3年的研究显示,在研究期间,DHEA-S水平低于54.5µg/dL的患者的死亡风险比较高水平的患者高64%111。
- 法国研究人员对290名受试者进行了为期10年的研究,发现DHEA-S水平低的男性的死亡风险比水平高的男性增加了1.9倍;在65-69岁的男性中尤其如此,他们的风险是6.5倍112。
- 在另一项对123名心脏病发作幸存者进行长达10年随访的研究中,发现低DHEA-S水平可预测心血管疾病导致的死亡113。
- 在参与法国一项基于社区的研究的622名65岁以上的老年人中,低DHEA-S水平与男性2年和4年内更高的死亡风险密切相关114。
- 在另一项研究中,当低DHEA-S与其他三种风险因素(睾酮缺乏、hs-CRP升高和血浆N-末端B型钠尿肽原升高)相结合时,心血管死亡风险比健康对照受试者高出惊人的63倍115。
有趣的是,除了DHEA-S的总体水平外,一些证据表明,它随着年龄的增长而下降的速度可能独立影响寿命。在一项针对950名65岁或65岁以上个体的研究中,那些DHEA-S水平下降幅度更大的受试者在研究期间死亡的可能性比DHEA-S下降幅度更慢的受试对象高75%。尽管本研究中基线DHEA-S与死亡率无关,但这些发现仍然存在116。
DHEA与癌症风险
由于DHEA可能会增加性激素水平,人们对其在患有或患有激素相关癌症的人中的使用表示担忧。迄今为止,没有任何研究可令人信服地表明,由于补充DHEA或孕烯醇酮,导致人类激素依赖性癌症风险增加1,7。
实际上,真相更加微妙。哈佛泌尿科医生Abraham Morgentaler和其他人的重要研究表明,低睾酮水平可能会增加前列腺癌的风险,尽管这是一个有争议的概念。Morgentaler本人已经成为在老年男性中补充睾酮的有力支持者。他还是一项研究的首席研究员,该研究表明,在大鼠中补充DHEA可以提高总睾酮水平,而不会对前列腺组织产生任何有害变化117。
类似的理论风险适用于乳腺癌。但是,在天然雌二醇和孕酮(DHEA和/或孕烯醇酮的天然产物)组合的大型研究中,没有证明患乳腺癌的风险增加。此外,单独使用天然孕酮可以降低患癌症风险。这再次表明,使用DHEA和孕烯醇酮等前体提高性激素水平是安全的。最近的一项动物研究证明了DHEA对肥胖大鼠的直接抗癌作用118。
任何已知患有任何类型癌症的个人在使用任何新的补充剂或药物(包括DHEA)前,应咨询其医生。
参考文献:
1. Traish AM et al. Dehydroepiandrosterone (DHEA)--a precursor steroid or an active hormone in human physiology. J Sex Med. 2011 Nov;8(11):2960-82; quiz 2983.
2. Savineau JP et al. Role of DHEA in cardiovascular diseases. Biochemical pharmacology. Mar 15 2013;85(6):718-726.
3. Samaras N et al. A review of age related dehydroepiandrosterone (DHEA) decline and its association with well-known geriatric syndromes. Is treatment beneficial? Rejuvenation research. 2013 Aug;16(4):285-94.
4. Dong Y et al. Dehydroepiandrosterone sulphate: action and mechanism in the brain. Journal of neuroendocrinology. Jan 2012;24(1):215-224.
5. Załuska M et al. [Dehydroepiandrosteron (DHEA) in the mechanisms of stress and depression]. Psychiatr Pol. 2009 May-Jun;43(3):263-74.
6. Straub RH et al. Replacement therapy with DHEA plus corticosteroids in patients with chronic inflammatory diseases--substitutes of adrenal and sex hormones. Zeitschrift fur Rheumatologie. 2000;59 Suppl 2:Ii/108-118.
7. Krysiak R et al. Current views on the role of dehydroepiandrosterone in physiology, pathology and therapy. Pol Merkur Lekarski. 2008 Jan;24(139):66-71.
8. Lopez-Marure R et al. Effects of dehydroepiandrosterone on proliferation, migration, and death of breast cancer cells. European journal of pharmacology. Jun 25 2011;660(2-3):268-274.
9. Arlt W et al. Oral dehydroepiandrosterone for adrenal androgen replacement: pharmacokinetics and peripheral conversion to androgens and estrogens in young healthy females after dexamethasone suppression. The Journal of clinical endocrinology and metabolism. Jun 1998;83(6):1928-1934.
10. Morales AJ et al. Effects of replacement dose of dehydroepiandrosterone in men and women of advancing age. J Clin Endocrinol Metab. 1994 Jun;78(6):1360-7.
11. Aldred S et al. Decreased dehydroepiandrosterone (DHEA) and dehydroepiandrosterone sulfate (DHEAS) concentrations in plasma of Alzheimer's disease (AD) patients. Arch Gerontol Geriatr. 2010 Jul-Aug;51(1):e16-8.
12. Panjari M et al. DHEA therapy for women: effect on sexual function and wellbeing. Human reproduction update. May-Jun 2007;13(3):239-248.
13. Schulze J et al. SULT2A1 Gene Copy Number Variation is Associated with Urinary Excretion Rate of Steroid Sulfates. Frontiers in endocrinology. 2013 Jul 12:4:88.
14. Fitzpatrick JL et al. Metabolism of DHEA by cytochromes P450 in rat and human liver microsomal fractions. Archives of biochemistry and biophysics. May 15 2001;389(2):278-287.
15. Miller KK et al. Stereo- and regioselectivity account for the diversity of dehydroepiandrosterone (DHEA) metabolites produced by liver microsomal cytochromes P450. Drug metabolism and disposition: the biological fate of chemicals. Mar 2004;32(3):305-313.
16. Holtorf K. The bioidentical hormone debate: are bioidentical hormones (estradiol, estriol, and progesterone) safer or more efficacious than commonly used synthetic versions in hormone replacement therapy? Postgrad Med. 2009;121(1): 73-85.
17. Zwain IH et al. Dehydroepiandrosterone: biosynthesis and metabolism in the brain. Endocrinology. 1999 Feb;140(2):880-7.
18. Liu D et al. Dehydroepiandrosterone activates endothelial cell nitric-oxide synthase by a specific plasma membrane receptor coupled to Galpha(i2,3). The Journal of biological chemistry. Jun 14 2002;277(24):21379-21388.
19. Liu D et al. Dehydroepiandrosterone stimulates nitric oxide release in vascular endothelial cells: evidence for a cell surface receptor. Steroids. Apr 2004;69(4):279-289.
20. Simoncini T et al. Dehydroepiandrosterone modulates endothelial nitric oxide synthesis via direct genomic and nongenomic mechanisms. Endocrinology. Aug 2003;144(8):3449-3455.
21. Lazaridis I et al. Neurosteroid dehydroepiandrosterone interacts with nerve growth factor (NGF) receptors, preventing neuronal apoptosis. PLoS biology. Apr 2011;9(4):e1001051.
22. Baulieu EE et al. Dehydroepiandrosterone (DHEA) and dehydroepiandrosterone sulfate (DHEAS) as neuroactive neurosteroids. Proceedings of the National Academy of Sciences of the United States of America. Apr 14 1998;95(8):4089-4091.
23. Charalampopoulos I et al. Neurosteroids as modulators of neurogenesis and neuronal survival. Trends Endocrinol Metab. 2008 Oct; 19(8):300-7.
24. Valenti G et al. Dehydroepiandrosterone and cognitive function in the elderly: The InCHIANTI Study. J Endocrinol Invest. 2009 Oct; 32(9):766-72.
25. Alhaj HA et al. Effects of DHEA administration on episodic memory, cortisol and mood in healthy young men: a double-blind, placebo-controlled study. Psychopharmacology (Berl). 2006 Nov; 188(4):541-51.
26. Stangl B et al. Administration of dehydroepiandrosterone (DHEA) enhances visual-spatial performance in postmenopausal women. Behav Neurosci. 2011 Oct;125(5):742-52.
27. Yamada S et al. Effects of dehydroepiandrosterone supplementation on cognitive function and activities of daily living in older women with mild to moderate cognitive impairment. Geriatr Gerontol Int. 2010 Oct;10(4):280-7.
28. Luppi C et al. Growth factors decrease in subjects with mild to moderate Alzheimer's disease (AD): potential correction with dehydroepiandrosterone-sulphate (DHEAS). Arch Gerontol Geriatr. 2009;49 Suppl 1:173-84.
29. Herbert J. Neurosteroids, brain damage, and mental illness. Exp Gerontol. 1998 Nov-Dec;33(7-8):713-27.
30. Wong SY et al. Low DHEAS levels are associated with depressive symptoms in elderly Chinese men: results from a large study. Asian J Androl. 2011 Nov;13(6):898-902.
31. Ritsner MS. The clinical and therapeutic potentials of dehydroepiandrosterone and pregnenolone in schizophrenia. Neuroscience. 2011 Sep 15; 191:91-100.
32. Strous RD et al. Hormonal profile effects following dehydroepiandrosterone (DHEA) administration to schizophrenic patients. Clin Neuropharmacol. 2005 Nov-Dec;28(6):265-9.
33. Bloch M et al. Dehydroepiandrosterone treatment of midlife dysthymia. Biol Psychiatry. 1999 Jun 15;45(12):1533-41.
34. Rabkin JG et al. Placebo-controlled trial of dehydroepiandrosterone (DHEA) for treatment of nonmajor depression in patients with HIV/AIDS. Am J Psychiatry. 2006 Jan; 163(1):59-66.
35. Brooke AM et al. Dehydroepiandrosterone improves psychological well-being in male and female hypopituitary patients on maintenance growth hormone replacement. J Clin Endocrinol Metab. 2006 Oct; 91(10):3773-9.
36. Cawthon PM. Gender Differences in Osteoporosis and Fractures. Clin Orthop Relat Res. 2011 Jul;469(7):1900-5
37. Kawate H et al. [Secondary osteoporosis UPDATE. Treatment of male osteoporosis. Testosterone replacement therapy etc]. Clin Calcium. 2010 May;20(5):744-51.
38. Nuti R et al. Bone fragility in men: where are we? J Endocrinol Invest. 2010;33(7 Suppl):33-8.
39. Fingerova H et al. [Reduced serum dehydroepiandrosterone levels in postmenopausal osteoporosis]. Ceska gynekologie / Ceska lekarska spolecnost J. Ev. Purkyne. Apr 1998;63(2):110-113.
40. Adachi M et al. Role of androgens and DHEA in bone metabolism, Clin Calcium 2006 Jan; 16(1):61-6.
41. Wang YD et al. Dehydroepiandrosterone indirectly inhibits human osteoclastic resorption via activating osteoblastic viability by the MAPK pathway. Chin Med J (Engl). 2012 Apr; 125(7):1230-5.
42. Corina M et al. Relationship between bone mineral density, weight, and estrogen levels in pre- and postmenopausal women. Rev Med Chir Soc Med Nat Iasi. 2012 Oct-Dec;116(4):946-50.
43. Weiss EP et al. Dehydroepiandrosterone replacement therapy in older adults: 1- and 2-y effects on bone. Am J Clin Nutr. 2009 May; 89(5): 1459–1467.
44. Okuno S et al. Serum levels of C-terminal telopeptide of type I collagen: a useful new marker of cortical bone loss in hemodialysis patients. Osteoporos Int. 2005 May;16(5):501-9.
45. von Mühlen D et al. Effect of dehydroepiandrosterone supplementation on bone mineral density, bone markers, and body composition in older adults: the DAWN trial. Osteoporos Int. 2008 May;19(5):699-707.
46. Shufelt C et al. DHEA-S levels and cardiovascular disease mortality in postmenopausal women: results from the National Institutes of Health--National Heart, Lung, and Blood Institute (NHLBI)-sponsored Women's Ischemia Syndrome Evaluation (WISE). J Clin Endocrinol Metab. 2010 Nov;95(11):4985-92.
47. Ponholzer A et al. Vascular risk factors and their association to serum androgen levels in a population-based cohort of 75-year-old men over 5 years: results of the VITA study. World J Urol. 2009 Apr;28(2):209-14.
48. Mitchell LE et al. Evidence for an association between dehydroepiandrosterone sulfate and nonfatal, premature myocardial infarction in males. Circulation. 1994 Jan; 89(1):89-93.
49. Martina V et al. Short-term dehydroepiandrosterone treatment increases platelet cGMP production in elderly male subjects. Clin Endocrinol (Oxf). 2006 Mar; 64(3):260-4.
50. Li Y et al. Dehydroepiandrosterone inhibits CD40/CD40L expression on human umbilical vein endothelial cells induced by interferon gamma. Int Immunopharmacol. 2009 Feb; 9(2):168-72
51. Gómez-Santos C et al. Differential effect of oral dehydroepiandrosterone-sulphate on metabolic syndrome features in pre- and postmenopausal obese women. Clin Endocrinol (Oxf). 2012 Oct;77(4):548-54.
52. Gómez-Santos C et al. Dehydroepiandrosterone-sulphate replacement improves the human plasma fatty acid profile in plasma of obese women. Steroids. Dec 11 2011;76(13):1425-1432.
53. Shufelt C et al. DHEA-S levels and cardiovascular disease mortality in postmenopausal women: results from the National Institutes of Health--National Heart, Lung, and Blood Institute (NHLBI)-sponsored Women's Ischemia Syndrome Evaluation (WISE). J Clin Endocrinol Metab. 2010 Nov;95(11):4985-92.
54. Dumas de la Roque E et al. Dehydroepiandrosterone: A new treatment for vascular remodeling diseases including pulmonary arterial hypertension. Pharmacology & therapeutics. May 2010;126(2):186-199.
55. Basat O et al. Visceral adipose tissue and an indicator of insulin resistance in nonobese patients with new onset type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes. 2006;114(2):58-62.
56. Steinberger J et al. Obesity, Insulin Resistance, Diabetes, and Cardiovascular Risk in Children. Circulation. 2003;107:1448-53.
57. Heurta-Garcia E et al. Dehydroepiandrosterone inhibits the activation and dysfunction of endothelial cells induced by high glucose concentration. 2012 Feb;v.77(3),p.233-240.
58. Weiss EP et al. Dehydroepiandrosterone (DHEA) replacement decreases insulin resistance and lowers inflammatory cytokines in aging humans. Aging (Albany NY). 2011 May; 3(5): 533–542.
59. Talaei A et al. The effect of dehydroepiandrosterone on insulin resistance in patients with impaired glucose tolerance. Hormones (Athens). 2010 Oct-Dec;9(4):326-31.
60. Dhatariya K et al. Effect of dehydroepiandrosterone replacement on insulin sensitivity and lipids in hypoadrenal women. Diabetes. 2005 Mar;54(3):765-9.
61. Ponikowska B et al. Gonadal and adrenal androgen deficiencies as independent predictors of increased cardiovascular mortality in men with type II diabetes mellitus and stable coronary artery disease. Int J Cardiol. 2010 Sep 3;143(3):343-8.
62. Brignardello E et al. Dehydroepiandrosterone administration counteracts oxidative imbalance and advanced glycation end product formation in type 2 diabetic patients. Diabetes Care. 2007 Nov; 30(11):2922-7.
63. Butcher SK et al. Raised cortisol: DHEAS ratios in the elderly after injury: potential impact upon neutrophil function and immunity. Aging cell. Dec 2005;4(6):319-324.
64. Buford TW et al. Impact of DHEA(S) and cortisol on immune function in aging: a brief review. Appl Physiol Nutr Metab. 2008 Jun;33(3):429-33.
65. Roxas M et al. Colds and influenza: a review of diagnosis and conventional, botanical, and nutritional considerations. Altern Med Rev. 2007 Mar;12(1):25-48.
66. Puizina-Ivić N et al. Modern approach to topical treatment of aging skin. Coll Antropol. 2010 Sep;34(3):1145-53.
67. Chan C et al. Effect of dehydroepiandrosterone on atopic dermatitis-like skin lesions induced by 1-chloro-2,4-dinitrobenzene in mouse. Journal of dermatological science. Nov 01 2013.
68. El-Alfy M et al. Skin responses to topical dehydroepiandrosterone: implications in antiageing treatment? Br J Dermatol. 2010 Nov;163(5):968-76.
69. Labrie F. DHEA, important source of sex steroids in men and even more in women. Prog Brain Res. 2010;182:97-148.
70. Calvo E et al. Pangenomic changes induced by DHEA in the skin of postmenopausal women. J Steroid Biochem Mol Biol. 2008 Dec; 112(4-5):186-93.
71. Nouveau S et al. Effects of topical DHEA on aging skin: a pilot study. Maturitas. 2008 Feb 20;59(2):174-81.
72. Yasui T et al. Androgen in postmenopausal women. J Med Invest. 2012;59(1-2):12-27.
73. Labrie F et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause. 2009 Sep-Oct; 16(5):923-31
74. Genazzani AR et al. Effect of 1-year, low-dose DHEA therapy on climacteric symptoms and female sexuality. Climacteric. 2011 Dec;14(6):661-8.
75. Reiter WJ et al. Dehydroepiandrosterone in the treatment of erectile dysfunction: a prospective, double-blind, randomized, placebo-controlled study. Urology. 1999 Mar;53(3):590-4; discussion 594-5.
76. Worrel ME et al. Effects of 7-keto dehydroepiandrosterone on voluntary ethanol intake in male rats. Alcohol (Fayetteville, N.Y.). Jun 2011;45(4):349-354.
77. Amato RJ et al. A comparison of dehydroepiandrosterone and 7-keto dehydroepiandrosterone with other drugs that modulate ethanol intake in rats responding under a multiple schedule. Behavioural pharmacology. Jun 2012;23(3):250-261.
78. Bobyleva V et al. The effects of the ergosteroid 7-oxo-dehydroepiandrosterone on mitochondrial membrane potential: possible relationship to thermogenesis. Archives of biochemistry and biophysics. May 1 1997;341(1):122-128.
79. Ihler G et al. 7-oxo-DHEA and Raynaud's phenomenon. Medical hypotheses. Mar 2003;60(3):391-397.
80. Hampl R et al. Steroids and thermogenesis. Physiol Res. 2006; 55(2):123-31.
81. Zenk JL et al. HUM5007, a novel combination of thermogenic compounds, and 3-acetyl-7-oxo-dehydroepiandrosterone: each increases the resting metabolic rate of overweight adults. J Nutr Biochem. 2007 Sep; 18(9):629-34.
82. Moyer AE et al. Stress-induced cortisol response and fat distribution in women. Obesity research. May 1994;2(3):255-262.
83. Abraham SB et al. Cortisol, obesity, and the metabolic syndrome: a cross-sectional study of obese subjects and review of the literature. Obesity (Silver Spring, Md.). Jan 2013;21(1):E105-117.
84. Muller C et al. The native anti-glucocorticoid paradigm. J Steroid Biochem Mol Biol. 2006 Jul; 100(1-3):95-105.
85. Hennebert O et al. Dehydroepiandrosterone 7alpha-hydroxylation in human tissues: possible interference with type 1 11beta-hydroxysteroid dehydrogenase-mediated processes. J Steroid Biochem Mol Biol. 2007 May;104(3-5):326-33.
86. Marin P et al. Cortisol secretion in relation to body fat distribution in obese premenopausal women. Metabolism: clinical and experimental. Aug 1992;41(8):882-886.
87. Buoso E et al. Opposing effects of cortisol and dehydroepiandrosterone on the expression of the receptor for Activated C Kinase 1: implications in immunosenescence. Experimental gerontology. Nov 2011;46(11):877-883.
88. Davidson M et al. Safety and pharmacokinetic study with escalating doses of 3-acetyl-7-oxo-dehydroepiandrosterone in healthy male volunteers. Clin Invest Med. 2000 Oct;23(5):300-10.
89. Ramos-Casals M et al. Autoimmunity and geriatrics: clinical significance of autoimmune manifestations in the elderly. Lupus. 2003;12(5):341-55.
90. Khorram O et al. Activation of immune function by dehydroepiandrosterone (DHEA) in age-advanced men. J Gerontol A Biol Sci Med Sci . 1997 Jan;52(1):M1-M7.
91. James K et al. IL-6, DHEA and the ageing process. Mech Ageing Dev . 1997 Feb;93(1-3):15-24.
92. Straub RH et al. Serum dehydroepiandrosterone (DHEA) and DHEA sulfate are negatively correlated with serum interleukin-6 (IL-6), and DHEA inhibits IL-6 secretion from mononuclear cells in man in vitro: possible link between endocrinosenescence and immunosenescence. J Clin Endocrinol Metab . 1998 Jun;83(6):2012-7.
93. Straub RH et al. Dehydroepiandrosterone in relation to other adrenal hormones during an acute inflammatory stressful disease state compared with chronic inflammatory disease: role of interleukin-6 and tumour necrosis factor. Eur J Endocrinol . 2002 Mar;146(3):365-74.
94. Leowattana W. DHEA(S): the fountain of youth. J Med Assoc Thai. 2001 Oct;84(Suppl 2):S605-S612.
95. Choi IS et al. Effects of dehydroepiandrosterone on Th2 cytokine production in peripheral blood mononuclear cells from asthmatics. Korean J Intern Med. 2008 Dec; 23(4):176-81.
96. Hazeldine J et al. Dehydroepiandrosterone as a regulator of immune cell function. J Steroid Biochem Mol Biol. 2010 May 31;120(2-3):127-36.
97. Drbalová K et al. Immunoprotective steroids and SHBG in non-treated hypothyroidism and their relationship to autoimmune thyroid disorders. Physiol Res. 2008;57 Suppl 1:S119-25.
98. Simunkova K et al. Adrenocortical function in young adults with diabetes mellitus type 1. J Steroid Biochem Mol Biol. 2010 Oct;122(1-3):35-41.
99. Crosbie D et al. Dehydroepiandrosterone for systemic lupus erythematosus. Cochrane Database Syst Rev. 2007 Oct 17;(4):CD005114.
100. van Vollenhoven RF et al. Treatment of systemic lupus erythematosus with dehydroepiandrosterone: 50 patients treated up to 12 months. J Rheumatol . 1998 Feb;25(2):285-9.
101. Chang DM et al. Dehydroepiandrosterone treatment of women with mild-to-moderate systemic lupus erythematosus: a multicenter randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2002 Nov;46(11):2924-7.
102. Sanders JL et al. Concurrent change in dehydroepiandrosterone sulfate and functional performance in the oldest old: results from the Cardiovascular Health Study All Stars study. The journals of gerontology. Series A, Biological sciences and medical sciences. Sep 2010;65(9):976-981.
103. Kenny AM et al. Dehydroepiandrosterone combined with exercise improves muscle strength and physical function in frail older women. J Am Geriatr Soc. 2010 Sep;58(9):1707-14.
104. Barrett-Connor E et al. A prospective study of dehydroepiandrosterone sulfate, mortality, and cardiovascular disease. The New England journal of medicine. Dec 11 1986;315(24):1519-1524.
105. Trivedi DP et al. Dehydroepiandrosterone sulfate and mortality in elderly men and women. The Journal of clinical endocrinology and metabolism. Sep 2001;86(9):4171-4177.
106. Hsu HJ et al. Low plasma DHEA-S increases mortality risk among male hemodialysis patients. Experimental gerontology. Dec 2012;47(12):950-957.
107. Kakiya R et al. Decreased serum adrenal androgen dehydroepiandrosterone sulfate and mortality in hemodialysis patients. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association. Oct 2012;27(10):3915-3922.
108. Ohlsson C et al. Low serum levels of dehydroepiandrosterone sulfate predict all-cause and cardiovascular mortality in elderly Swedish men. The Journal of clinical endocrinology and metabolism. Sep 2010;95(9):4406-4414.
109. Phillips AC et al. Cortisol, DHEA sulphate, their ratio, and all-cause and cause-specific mortality in the Vietnam Experience Study. European journal of endocrinology. Aug 2010;163(2):285-292.
110. Enomoto M et al. Serum dehydroepiandrosterone sulfate levels predict longevity in men: 27-year follow-up study in a community-based cohort (Tanushimaru study). Journal of the American Geriatrics Society. Jun 2008;56(6):994-998.
111. Glei DA et al. Dehydroepiandrosterone sulfate (DHEAS) and risk for mortality among older Taiwanese. Annals of epidemiology. Jul 2006;16(7):510-515.
112. Mazat L et al. Prospective measurements of dehydroepiandrosterone sulfate in a cohort of elderly subjects: relationship to gender, subjective health, smoking habits, and 10-year mortality. Proceedings of the National Academy of Sciences of the United States of America. Jul 3 2001;98(14):8145-8150.
113. Jansson JH et al. von Willebrand factor, tissue plasminogen activator, and dehydroepiandrosterone sulphate predict cardiovascular death in a 10 year follow up of survivors of acute myocardial infarction. Heart (British Cardiac Society). Oct 1998;80(4):334-337.
114. Berr C et al. Relationships of dehydroepiandrosterone sulfate in the elderly with functional, psychological, and mental status, and short-term mortality: a French community-based study. Proceedings of the National Academy of Sciences of the United States of America. Nov 12 1996;93(23):13410-13415.
115. Ponikowska B et al. Gonadal and adrenal androgen deficiencies as independent predictors of increased cardiovascular mortality in men with type II diabetes mellitus and stable coronary artery disease. Int J Cardiol. 2010 Sep 3;143(3):343-8.
116. Cappola AR et al. Trajectories of dehydroepiandrosterone sulfate predict mortality in older adults: the cardiovascular health study. The journals of gerontology. Series A, Biological sciences and medical sciences. Dec 2009;64(12):1268-1274.
117. Rhoden EL et al. Effects of chronic administration of dehydroepiandrosterone on serum testosterone levels and prostatic tissue in rats. The Journal of urology. Nov 2003;170(5):2101-2103.
118. Hakkak R et al. Dehydroepiandrosterone intake protects against 7,12-dimethylbenz(a)anthracene-induced mammary tumor development in the obese Zucker rat model. Oncology reports. Aug 2010;24(2):357-362.

